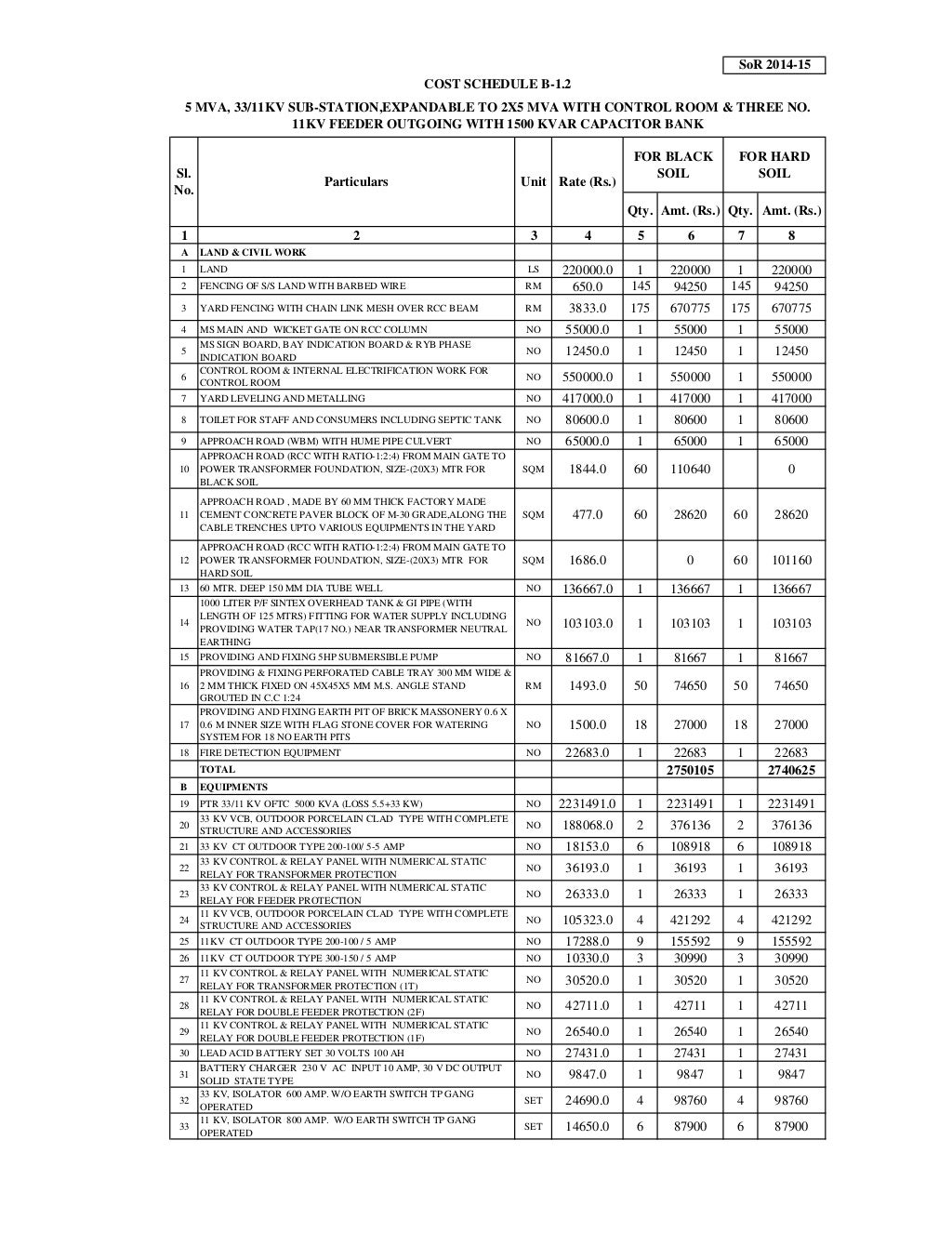
Medical Devices Regulations Canada Sor 98-282 . 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not required to be. Html (accessibility buttons available) |. Go to the justice canada site. this document outlines the strategy and provides guidance for the medical device industry on health. Type medical devices regulations in the title. 2 are amended by adding the following after section 2: 83 the medical devices regulations. how to find the medical devices regulations : regulatory agency means a government agency or other entity outside canada that has a legal right to control the.
from www.slideshare.net
Html (accessibility buttons available) |. Type medical devices regulations in the title. 2 are amended by adding the following after section 2: 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not required to be. this document outlines the strategy and provides guidance for the medical device industry on health. Go to the justice canada site. how to find the medical devices regulations : regulatory agency means a government agency or other entity outside canada that has a legal right to control the. 83 the medical devices regulations.
Sor
Medical Devices Regulations Canada Sor 98-282 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not required to be. Type medical devices regulations in the title. this document outlines the strategy and provides guidance for the medical device industry on health. 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not required to be. Html (accessibility buttons available) |. regulatory agency means a government agency or other entity outside canada that has a legal right to control the. how to find the medical devices regulations : 2 are amended by adding the following after section 2: Go to the justice canada site. 83 the medical devices regulations.
From www.slideshare.net
Sor Medical Devices Regulations Canada Sor 98-282 regulatory agency means a government agency or other entity outside canada that has a legal right to control the. Go to the justice canada site. Html (accessibility buttons available) |. 2 are amended by adding the following after section 2: Type medical devices regulations in the title. 83 the medical devices regulations. 95 (1) a medical. Medical Devices Regulations Canada Sor 98-282.
From www.pdffiller.com
Fillable Online Medical Devices Regulations ( SOR /98282) Fax Email Medical Devices Regulations Canada Sor 98-282 Type medical devices regulations in the title. how to find the medical devices regulations : 2 are amended by adding the following after section 2: this document outlines the strategy and provides guidance for the medical device industry on health. Go to the justice canada site. Html (accessibility buttons available) |. 95 (1) a medical device. Medical Devices Regulations Canada Sor 98-282.
From zhuanlan.zhihu.com
医疗器械出口加拿大MDEL、MDL 知乎 Medical Devices Regulations Canada Sor 98-282 83 the medical devices regulations. how to find the medical devices regulations : this document outlines the strategy and provides guidance for the medical device industry on health. 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not required to be. 2 are amended. Medical Devices Regulations Canada Sor 98-282.
From www.researchgate.net
(PDF) Current medical device regulation Is that enough? Medical Devices Regulations Canada Sor 98-282 Go to the justice canada site. how to find the medical devices regulations : regulatory agency means a government agency or other entity outside canada that has a legal right to control the. this document outlines the strategy and provides guidance for the medical device industry on health. 2 are amended by adding the following after. Medical Devices Regulations Canada Sor 98-282.
From www.nsf.org
Key FDA Policies for Medical Device Manufacturer Inspections NSF Medical Devices Regulations Canada Sor 98-282 Type medical devices regulations in the title. how to find the medical devices regulations : 2 are amended by adding the following after section 2: Go to the justice canada site. 83 the medical devices regulations. 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations. Medical Devices Regulations Canada Sor 98-282.
From www.hsiner.com
Hsiner has obtained MDSAP certificate Medical Devices Regulations Canada Sor 98-282 Go to the justice canada site. 2 are amended by adding the following after section 2: this document outlines the strategy and provides guidance for the medical device industry on health. 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not required to be. Html. Medical Devices Regulations Canada Sor 98-282.
From www.slideshare.net
Regulatory oversight of testing in Canada Health Canada pers… Medical Devices Regulations Canada Sor 98-282 Go to the justice canada site. how to find the medical devices regulations : Html (accessibility buttons available) |. 83 the medical devices regulations. regulatory agency means a government agency or other entity outside canada that has a legal right to control the. Type medical devices regulations in the title. 95 (1) a medical device. Medical Devices Regulations Canada Sor 98-282.
From www.isct-cytotherapy.org
Current state of Health Canada regulation for cellular and gene therapy Medical Devices Regulations Canada Sor 98-282 how to find the medical devices regulations : Html (accessibility buttons available) |. 83 the medical devices regulations. Go to the justice canada site. 2 are amended by adding the following after section 2: regulatory agency means a government agency or other entity outside canada that has a legal right to control the. this document. Medical Devices Regulations Canada Sor 98-282.
From www.presentationeze.com
Medical Device Approval European Regulatory ProcessPresentationEZE Medical Devices Regulations Canada Sor 98-282 83 the medical devices regulations. 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not required to be. Type medical devices regulations in the title. 2 are amended by adding the following after section 2: how to find the medical devices regulations : regulatory. Medical Devices Regulations Canada Sor 98-282.
From www.greenlight.guru
Ultimate Guide to 21 CFR Part 820 — FDA's Quality System Regulation Medical Devices Regulations Canada Sor 98-282 this document outlines the strategy and provides guidance for the medical device industry on health. 83 the medical devices regulations. regulatory agency means a government agency or other entity outside canada that has a legal right to control the. Type medical devices regulations in the title. Html (accessibility buttons available) |. how to find the medical. Medical Devices Regulations Canada Sor 98-282.
From www.regdesk.co
Important Things to Know Before Registering Your Medical Device in Medical Devices Regulations Canada Sor 98-282 Html (accessibility buttons available) |. regulatory agency means a government agency or other entity outside canada that has a legal right to control the. 2 are amended by adding the following after section 2: this document outlines the strategy and provides guidance for the medical device industry on health. Go to the justice canada site. 95. Medical Devices Regulations Canada Sor 98-282.
From learn.marsdd.com
Medical device regulations, classification & submissions Canada, US, EU Medical Devices Regulations Canada Sor 98-282 Html (accessibility buttons available) |. 2 are amended by adding the following after section 2: 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not required to be. Go to the justice canada site. this document outlines the strategy and provides guidance for the medical. Medical Devices Regulations Canada Sor 98-282.
From www.presentationeze.com
Differences between Australian & EU Medical Device Medical Devices Regulations Canada Sor 98-282 Go to the justice canada site. 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not required to be. Html (accessibility buttons available) |. this document outlines the strategy and provides guidance for the medical device industry on health. regulatory agency means a government. Medical Devices Regulations Canada Sor 98-282.
From www.tuvsud.com
EU In Vitro Diagnostic Medical Device Regulation TÜV SÜD Medical Devices Regulations Canada Sor 98-282 Go to the justice canada site. 2 are amended by adding the following after section 2: regulatory agency means a government agency or other entity outside canada that has a legal right to control the. Html (accessibility buttons available) |. 83 the medical devices regulations. this document outlines the strategy and provides guidance for the medical. Medical Devices Regulations Canada Sor 98-282.
From www.gbu-taganskij.ru
Regulations Of Medical Devices In Regulated Countries A, 41 OFF Medical Devices Regulations Canada Sor 98-282 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not required to be. 83 the medical devices regulations. Go to the justice canada site. Type medical devices regulations in the title. how to find the medical devices regulations : regulatory agency means a. Medical Devices Regulations Canada Sor 98-282.
From d417bradfordnguyen.blogspot.com
D Bradford Nguyen Health Canada Medical Device Recognized Standards Medical Devices Regulations Canada Sor 98-282 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not required to be. Go to the justice canada site. Html (accessibility buttons available) |. how to find the medical devices regulations : 2 are amended by adding the following after section 2: Type medical devices. Medical Devices Regulations Canada Sor 98-282.
From billalaser.weebly.com
Iso 13485 standard billalaser Medical Devices Regulations Canada Sor 98-282 Type medical devices regulations in the title. 2 are amended by adding the following after section 2: regulatory agency means a government agency or other entity outside canada that has a legal right to control the. 95 (1) a medical device that, on june 30, 1998, is being sold in canada pursuant to the old regulations is not. Medical Devices Regulations Canada Sor 98-282.
From learn.marsdd.com
Medical device regulations, classification & submissions Canada, US, EU Medical Devices Regulations Canada Sor 98-282 2 are amended by adding the following after section 2: this document outlines the strategy and provides guidance for the medical device industry on health. Type medical devices regulations in the title. Html (accessibility buttons available) |. 83 the medical devices regulations. regulatory agency means a government agency or other entity outside canada that has a legal. Medical Devices Regulations Canada Sor 98-282.